
Part 11 Is Not a Software Label
Most teams still treat Part 11 as a feature of software. It is not.
21 CFR Part 11 defines when electronic records and electronic signatures are considered reliable and equivalent to paper-based documentation. The regulation is not about tools. It is about control.
The key point is scope. Part 11 applies only when records are created, modified, maintained, or submitted under FDA predicate rules. A digital PSMF system does not automatically fall under Part 11 just because it is electronic. But once it supports regulated records, approvals, or signatures, expectations change.
Inspectors are not interested in the platform. They are interested in whether the system can demonstrate who made a change, what changed, when it changed, and why it changed. Platforms like PSMF Manager are built to make this level of traceability consistent and inspection-ready.
Why Digital PSMF Systems Sit at a Regulatory Intersection
The PSMF is governed by EU pharmacovigilance requirements, while Part 11 governs electronic records in FDA-regulated environments. This creates a clear overlap.
EMA GVP Module II requires the PSMF to describe the systems used to manage pharmacovigilance data, including validation status, data handling, and change control. If those systems support regulated electronic records, Part 11 expectations apply.
There is also an operational pressure. The PSMF must be provided within seven calendar days of a request. Systems that rely on manual compilation or disconnected records struggle to meet this consistently, especially when global updates are involved. This is why digital PSMF systems are now central to inspection readiness, not just efficiency.
For example, managing consistency across markets becomes easier with features like global and local PSMFs, where updates can be controlled centrally without duplication.
What Part 11 Compliance Actually Means in Practice
Part 11 is often reduced to audit trails, but compliance comes from a combination of controls working together.
| Control Area | What It Means in Practice | What Inspectors Look For |
|---|---|---|
| Validation | The system is proven fit for intended use with documented evidence | Clear validation documentation aligned with actual workflows and system usage |
| Audit Trail Integrity | Secure, time-stamped, non-editable logs of all changes | Who, what, when, and why the change was made |
| Access Control | Role-based permissions with no shared or uncontrolled access | Clear user attribution and restricted access based on roles |
| Electronic Signatures | Sign-offs uniquely linked to users and specific record versions | Evidence that approvals are tied to exact versions and cannot be reassigned |
| Record Retention & Retrieval | Records remain complete, readable, and accessible over time | Ability to reconstruct full history, including past versions and approvals |
EMA GVP Module II also expects validation status and system controls to be reflected within the PSMF, reinforcing the need for alignment between system behavior and documentation.
Features like tracked changes help ensure this level of visibility is consistently maintained.
How ALCOA+ Connects to Digital PSMF Systems
Regulators often assess data integrity using ALCOA+ principles. These are not separate from Part 11. They reflect how its expectations are applied in practice.
A well-controlled digital PSMF system ensures records are attributable through user-specific access, contemporaneous through time-stamped entries, and accurate through validated workflows. It also ensures records remain complete, consistent, and available throughout their lifecycle.
This is what inspectors expect to see. Not just a compliant system on paper, but one that consistently produces reliable, traceable records.

Where Compliance Breaks Down in Practice
Most failures do not come from missing features. They come from weak implementation.
A common pattern is treating the system as a document repository rather than a controlled environment. Versioning may happen outside the system, approvals may occur over email, and audit trails may exist but lack context such as the reason for change.
Another frequent issue is the absence of procedural control. Even a well-configured system fails if there are no clear SOPs governing its use or if training records are incomplete. Part 11 is as much about process as it is about technology.
There is also often a gap between what the PSMF describes and how the system actually operates. If the PSMF states that processes are controlled within a system, but in reality depend on manual workarounds, inspectors will focus on that mismatch.
The Role of QPPV in a Digital Environment
The QPPV is responsible for oversight of the pharmacovigilance system. Inspectors do not assess this through titles. They assess it through evidence.
Digital systems make this evidence visible. Review logs, approval records, and access to system-level information show whether the QPPV is actively involved. If the QPPV is named but not connected to system activity, it weakens the credibility of oversight.
Validation Remains the Hardest Layer
Validation is often misunderstood because it is treated as a one-time activity. In reality, it is about ongoing assurance that the system performs as intended.
There is no single prescribed method here. However, the expectation is clear. The organization must demonstrate that the system supports its workflows, maintains data integrity, and operates under control.
With increasing use of automation, including AI-supported features, this expectation extends further. There must be clarity on how automated functions behave and how they are governed.
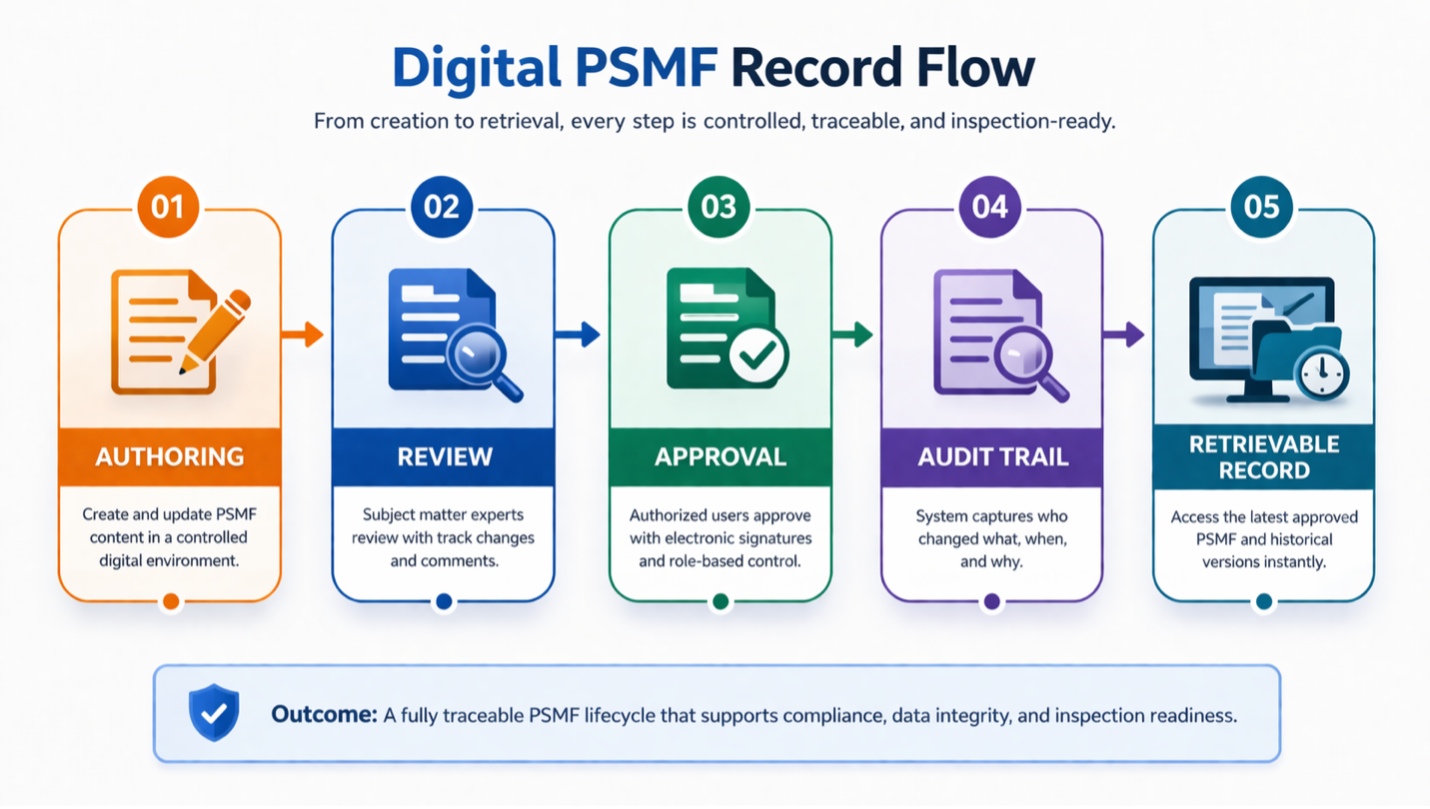
What Good Compliance Looks Like
A compliant digital PSMF system is defined by control.
There is a single source of truth. Access is role-based. Changes are traceable and reviewable. Approvals are controlled within the system. Historical versions are preserved. Most importantly, the PSMF reflects how the system is actually used.
Capabilities like automated PSMF generation further ensure that updated, controlled documents can be produced quickly without compromising traceability.
When these elements are in place, compliance becomes demonstrable. The system itself provides the evidence.
How to Assess Your Current System
Start by identifying whether your digital PSMF system supports records that fall under Part 11 scope. Then review whether validation is properly documented and aligned with actual system use.
Look at audit trails. Do they capture not just actions, but also the reasoning behind changes? Check whether access is controlled and whether user actions are clearly attributable.
Finally, compare the PSMF description with how the system actually operates. Any gap between the two is a potential inspection finding.
You can explore how a structured system approach works in practice on the PSMF Manager platform.
What 21 CFR Part 11 Compliance Really Means for Digital PSMF Systems
21 CFR Part 11 compliance is not about adopting a digital system. It is about demonstrating control over electronic records and signatures.
A digital PSMF system should do more than improve efficiency. It should provide clear, traceable evidence of how records are created, reviewed, approved, and maintained.
That is what inspectors evaluate.
And that is what compliance really means.
Request a demo to see how a system-driven approach supports validated workflows, traceable approvals, and inspection-ready PSMF management.