
Audits & Inspections Don’t Test Effort. They Test Control.
Most teams assume PSMF failures happen because of time pressure or lack of coordination.
That is rarely the real issue.
A review of the data related to recent pharmacovigilance inspections has identified over 100 findings, with the PSMF referenced in a significant portion of them. More importantly, regulatory inspection trends (EMA and MHRA PV inspection metrics) indicate that many major findings are linked to pharmacovigilance system governance gaps rather than case processing errors.
This changes how the problem should be understood.
Inspectors are not evaluating how much effort went into preparing the pharmacovigilance documentation seems more apt here than PSMF. They are evaluating whether the system was under control before the inspection began.
And this is exactly where manual PSMF management begins to fail.
When PSMF Timing Becomes a Compliance Risk
One of the most critical pressure points during inspections is the 7-day PSMF request requirement.
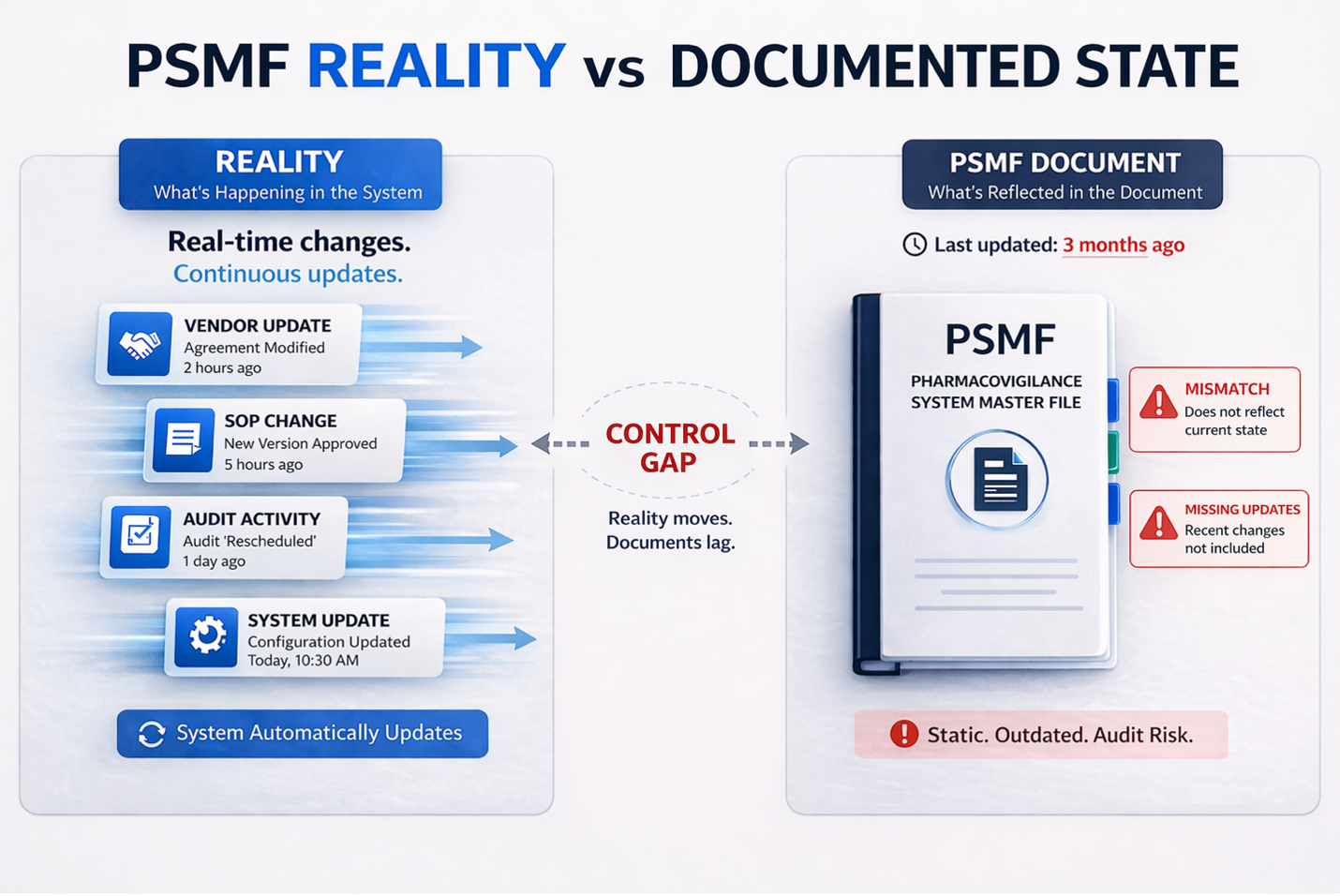
When regulators request the PSMF, it must reflect the system as it exists at that exact moment.
Manual systems typically respond in reactive ways. Teams attempt to update documents after the request or coordinate last-minute corrections across functions. This creates inconsistencies between the PSMF sections and annexes.
Inspectors are trained to detect this pattern. When updates appear reactive rather than continuous, it signals that the system was not in a state of control.
In many cases, this alone is enough to escalate findings.
The Three Failure Patterns Consistently Flagged
PSMF findings typically fall into three clear categories defined by regulatory expectations.
| Failure Type | What They See | What It Signals |
|---|---|---|
| Inadequate system description | Narrative does not match real workflows | Weak system understanding |
| Outdated or incorrect info | Annexes not aligned with current data | Poor data governance |
| Missing required content | Sections incomplete | Structural non-compliance |
These are not just Quality control issues. They indicate that the pharmacovigilance system is not being actively governed.
What This Looks Like in a Real Inspection
In one inspection scenario, regulators identified that Annex I did not reflect recent system changes. The logbook had been updated retrospectively, and several updates were missing entirely.
At the same time, access to a qualified medical professional was not clearly documented, and the ICSR workflow did not match actual operations.
The CAPA required restructuring the logbook process, redefining oversight responsibilities, and rebuilding documentation to reflect real workflows.
This was not treated as a documentation issue. It was treated as a governance failure.
Where Manual PSMF Management Breaks Down
Manual systems are not designed for continuously evolving pharmacovigilance environments.
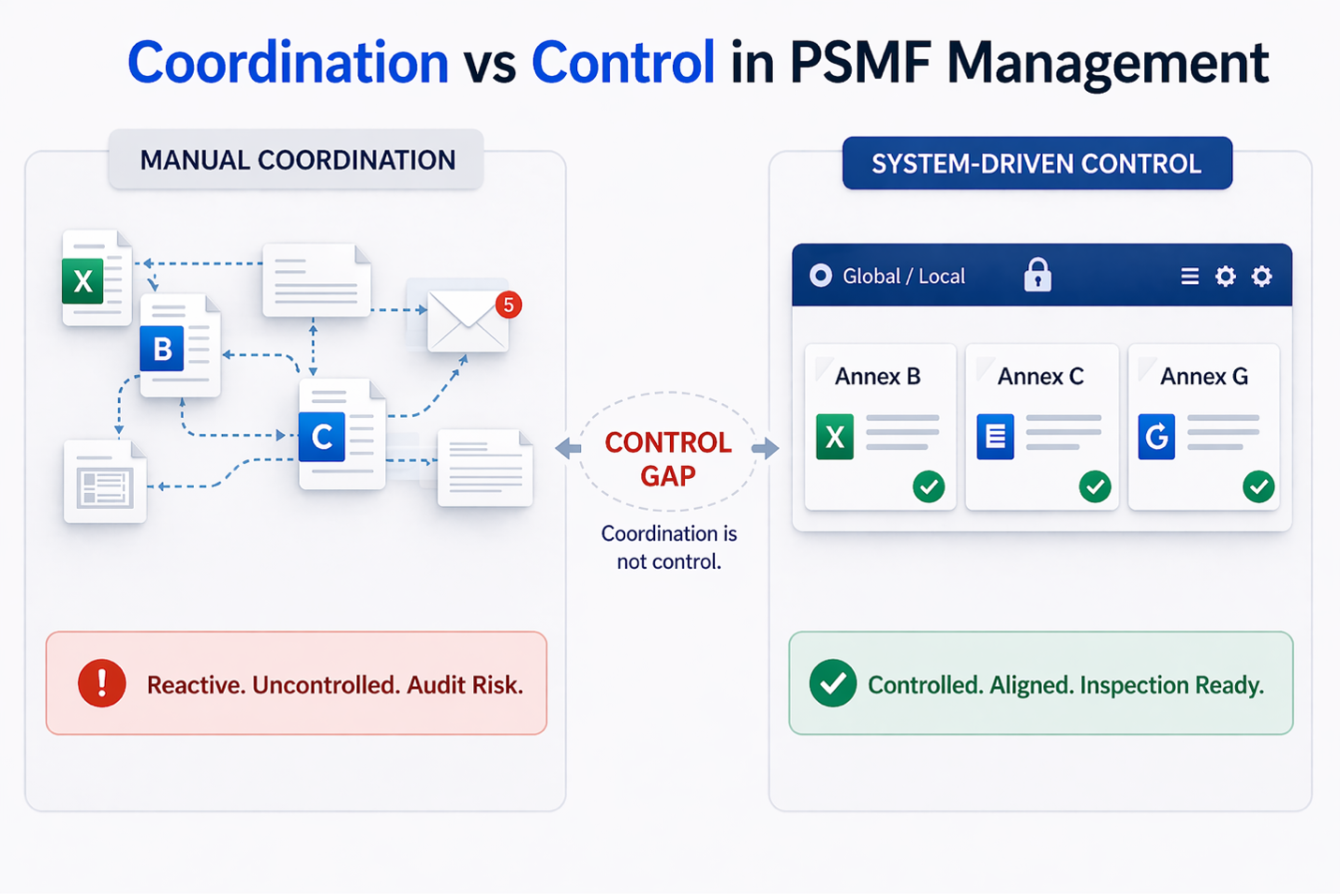
The breakdown typically begins with the absence of a single source of truth. Different teams maintain different versions, and alignment depends on communication rather than structure.
Traceability becomes difficult because updates are scattered across emails, spreadsheets, and local files. Establishing who made a change, when it was made, and whether it was approved becomes time-consuming.
Annexes create additional complexity. Vendor agreements, safety data sources, audit schedules, and change logs are frequently updated, but manual processes struggle to keep them aligned.
At scale, inconsistencies are not rare. They are expected.
Annex I Is Where Oversight Gets Tested
Among all sections of the pharmacovigilance system master file, Annex I receives the most scrutiny.
It serves as the chronological proof of system control.
When Annex I is incomplete or delayed, it raises immediate concerns about QPPV oversight. Even small gaps in the individual entries in the Change Log can weaken the credibility of the entire system.
Manual tracking often leads to missed entries, delayed updates, and a lack of context retrospectively. Over time, this creates a pattern where the logbook no longer reflects reality.
This gap is exactly where AI-driven Change Log automation comes in. Instead of relying on manual entries, AI can auto-generate Annex I Change Log updates at the time of change request creation, ensuring nothing is missed. It also reviews changes for accuracy, checks alignment with source data, and flags inconsistencies against EMA GVP Module II expectations. This shifts the Change Log from a retrospective activity to a real-time, system-driven record of control.
From an inspector’s perspective, this is not a minor issue. It is a breakdown in continuous oversight.
Global and Local PSMFs Often Fall Out of Sync
For organizations operating across regions, maintaining consistency between global and local PSMFs is a major challenge.
A global SOP may be updated centrally, but local PSMFs may not reflect that change immediately. This creates a period where different versions of the system exist simultaneously.
During inspections, these inconsistencies are easily identified.
A structured approach to global and local PSMF management helps ensure that updates are consistently reflected across all levels without relying on manual coordination.
Moving From Documents to Systems
The limitation of manual PSMF management is not the effort. It is the operating model.
Manual processes treat the PSMF as a document that needs periodic updates. Modern pharmacovigilance systems require it to function as a continuously updated representation of pharmacovigilance operations.
This shift introduces a different approach.
Instead of compiling information, organizations maintain structured systems where updates are recorded as they happen. Changes become visible immediately, and traceability is built into the process. Tracked changes provide visibility into updates without manual comparison. Integration with external data sources ensures that annexes reflect actual system inputs rather than manually entered data.
At the final stage, PSMF generation is no longer a compilation exercise. It becomes a direct output of structured, current data.
Why This Matters for the QPPV
Responsibility for the pharmacovigilance system does not stay with OR rely on with the document. It sits with the system owner.
QPPVs are expected to demonstrate continuous pharmacovigilance oversight. When inconsistencies appear in the PSMF, the concern is not who updated the file.
The concern is whether the system was under control.
This introduces a level of professional accountability that manual systems struggle to support.
The Difference Between Passing and Failing Passing and Failure
Teams relying on manual processes to prepare for inspections.
Teams using system-driven approaches always remain prepared.
That difference reflects the shift from reactive updates to continuous control.
And that is ultimately what auditors and inspectors evaluate.
Move from manual coordination to a system built for continuous compliance and inspection readiness. Request a demo